Arrangement of Electrons in the Atom
Bohr's Theory
The main points in Bohr's explanation of the fact that each element has a unique emission line spectrum are as follows:
The fact that a line spectrum is observed - and not a continuous one - shows that only specific energy transitions are possible within an atom. This is strong evidence for the existence of energy levels.
The photon of light emitted is of a particular frequency and need not be in the visible range. If electrons jump back to the first energy level it emits ultraviolet light (Lyman series) which is invisible. If electrons jump back to the third energy level it emits infared light (Paschen series) which is also invisible. However if an electron jumps back to the second energy level it emits visible light (Balmers series) which is what we see on the line spectrum. Jumps from different energy levels to the second energy level releases light of different colours.
Each element has a unique emission line spectrum because each element has a different number of electrons and each element has its own arrangement of these electrons in different energy levels. As a result, the different electrons transitions give rise to different emission spectra
Flame Test (Evidence for Bohr's Theory)
Element Colour
Lithium Crimson
Sodium Yellow
Potassium Lilac
Barium Green
Copper Blue-Green
Strontium Red
Limitations of Bohr's Theory:
- Electrons in the ground state have energy of fixed values i.e. the energy of the electrons in a particular orbit or energy level is quantised
- If energy is provided to an atom in its ground state, a specific amount of this energy is absorbed and the electrons jump to higher energy levels i.e. an excited state
- This excited state is unstable and the electrons fall back to lower energy levels
- As electrons fall back, the excess energy is released in the form of light of a definite amount of energy. The frequency of the light depends on the difference in energy levels and is given by the equation E2 - E1 = hf (h is plancks constant, f is the frequency)
- A line spectrum is observed
The fact that a line spectrum is observed - and not a continuous one - shows that only specific energy transitions are possible within an atom. This is strong evidence for the existence of energy levels.
The photon of light emitted is of a particular frequency and need not be in the visible range. If electrons jump back to the first energy level it emits ultraviolet light (Lyman series) which is invisible. If electrons jump back to the third energy level it emits infared light (Paschen series) which is also invisible. However if an electron jumps back to the second energy level it emits visible light (Balmers series) which is what we see on the line spectrum. Jumps from different energy levels to the second energy level releases light of different colours.
Each element has a unique emission line spectrum because each element has a different number of electrons and each element has its own arrangement of these electrons in different energy levels. As a result, the different electrons transitions give rise to different emission spectra
Flame Test (Evidence for Bohr's Theory)
- Place a sample of the salt on a damp wooden splint
- Place it in the bunsen burner and note the colour the salt burns
- The colours imparted by different salts are as follows
Element Colour
Lithium Crimson
Sodium Yellow
Potassium Lilac
Barium Green
Copper Blue-Green
Strontium Red
Limitations of Bohr's Theory:
- Bohr's Theory only worked to explain the emission spectrum of hydrogen. When his theory was applied to atoms with more than on electron, it failed to account for many of the lines in the emission spectra of these atoms
- It was found that electrons do not travel in a fixed path, a fixed distance from the nucleus (Heisenberg Uncertainty Principle)
Atomic Absorption Spectrometry
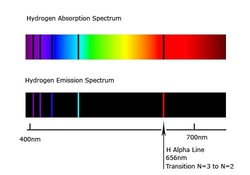
In Atomic Absorption Spectrometry, white light is passed through a gaseous sample of the element, and will have specific frequencies removed from it. Whereas emission spectrum is coloured lines on a black background, absorption spectrum is black lines on a coloured background.
This technique is used to determine the concentration of elements
Emission Spectra Atomic Absorption Spectra
1) Identifying Elements 1) Identifying Elements
2) Sodium street lamps 2) Concentration of Elements
3) Giving particular colour to fireworks 3) Concentration of heavy metals in water
This technique is used to determine the concentration of elements
Emission Spectra Atomic Absorption Spectra
1) Identifying Elements 1) Identifying Elements
2) Sodium street lamps 2) Concentration of Elements
3) Giving particular colour to fireworks 3) Concentration of heavy metals in water
Energy Levels
ENERGY LEVEL: fixed energy value an electron in an atom may have
Inside an atom, the electron occupy only certain positions with a very definite radius from the nucleus (energy levels, orbits, shells). An electron occupying these energy levels can only possess very specific amounts of energy.
To determine how many electrons each energy level can hold we use the formula 2n² (n is the number of the energy level)
Inside an atom, the electron occupy only certain positions with a very definite radius from the nucleus (energy levels, orbits, shells). An electron occupying these energy levels can only possess very specific amounts of energy.
To determine how many electrons each energy level can hold we use the formula 2n² (n is the number of the energy level)
Energy Sub-Levels
On closer examination, the bands of light in an emission spectrum can be seen to consist of two or more lines very close together. This suggests that a main energy level may be made up of parts of slightly different energy known as energy sub levels which are denoted by the letters s, p and d
s sub-level (holds two electrons)
p sub-level (holds six electrons)
d sub-level (holds ten electrons)
s sub-level (holds two electrons)
p sub-level (holds six electrons)
d sub-level (holds ten electrons)
Wave Nature of the Electron
Although electrons were identified as particles it can also be seen to possess properties associated with waves e.g. shown to have a wavelength. This means they do not travel in a fixed bath at a definite distance from the nucleus
HEISENBERG UNCERTAINTY PRINCIPLE: it is impossible to measure at the same time both the velocity and the position of an electron
HEISENBERG UNCERTAINTY PRINCIPLE: it is impossible to measure at the same time both the velocity and the position of an electron
- In order to determine location or velocity of an electron within an atom, it must be illuminated with radiation
- However radiation interferes with the electron by changing the position and/or momentum of the electron
- This means radiation is giving 'false' information
Atomic Orbitals
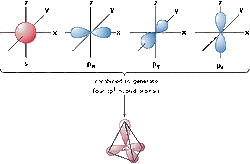
AN ORBITAL: is a region in space within which there is a high probability of finding an electron
Erwin Schrodinger used mathematics to calculate regions where an electron is likely to be found. He called them atomic Orbitals
There is one s orbitals is each energy level and they are spherical
There are three p orbitals in an energy level - px, py and pz. They are arranged at right angles to each other
Erwin Schrodinger used mathematics to calculate regions where an electron is likely to be found. He called them atomic Orbitals
There is one s orbitals is each energy level and they are spherical
There are three p orbitals in an energy level - px, py and pz. They are arranged at right angles to each other

