Chemical Bonding: Chemical Formulas
Compounds
COMPOUND: is a substance that is made up of two or more different elements combined together chemically
The noble gases do not tend to form compounds and so their electronic configurations are taken to be particularly stable
The noble gases do not tend to form compounds and so their electronic configurations are taken to be particularly stable
- Argon is used in light bulbs to prevent the filament from burning
- Helium is used in balloons
The Octet Rule
THE OCTET RULE: states that when bonding occurs, atoms tend to reach an electron arrangement with eight electrons in the outermost shell
Not a strict rule as there is a number of exceptions to it
Not a strict rule as there is a number of exceptions to it
- Having formed a compound, transition elements do not necessarily have eight electrons in their outer shell
- Having formed a compound elements near helium tend to have only two electrons in their outer shell
Ionic Bonding and Ionic Compounds
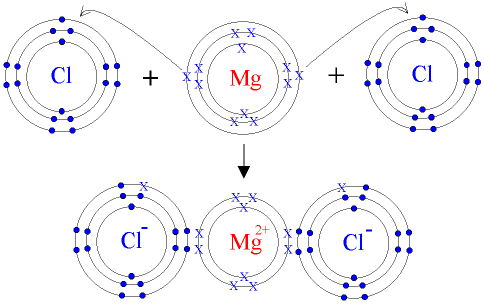
IONIC BOND: is the force of attraction between two oppositely charged ions in a compound
Is formed when electrons are transferred from one atom to another resulting in ion formation. Ion achieves electronic configuration of the nearest noble gas
Is formed when electrons are transferred from one atom to another resulting in ion formation. Ion achieves electronic configuration of the nearest noble gas
Crystal Lattices: Not correct to speak of a molecule of an ionic compound. Force of attraction of the charge on an ion is distributed in 3D manner around ion. Results in formation of crystal lattice i.e. in sodium chloride every sodium ion is surrounded by six chloride ions and vice versa
d-Block and Transistion Elements
TRANSITION ELEMENTS: is one that forms at least one ion with a partially filled d sub-level
As the 3p and 4s sub-level are close in energy, this results in transition metals losing varying number of electrons (variable valency)
Characteristic properties of transition metals
i) Neither scandium nor zinc exhibit these properties and strictly speaking are not transition metals and are more correctly classified as d-block elements
ii) When transition metals react, the first electron is lost from the 4s sub-level as this is part of the outer shell
As the 3p and 4s sub-level are close in energy, this results in transition metals losing varying number of electrons (variable valency)
Characteristic properties of transition metals
- The exhibit variable valency
- They form coloured compounds
- They can behave as catalysts
i) Neither scandium nor zinc exhibit these properties and strictly speaking are not transition metals and are more correctly classified as d-block elements
ii) When transition metals react, the first electron is lost from the 4s sub-level as this is part of the outer shell
Covalent Bonding
COVALENT BOND: formed when two atoms share a pair of electrons
VALENCY: the number of atoms of hydrogen or any other monovalent element with which each atom of that element combines
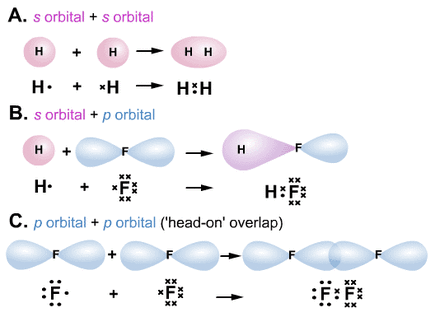
Sigma and Pi Bonding:
VALENCY: the number of atoms of hydrogen or any other monovalent element with which each atom of that element combines
Sigma and Pi Bonding:
|
Sigma Bonding:
A. s orbital --- s orbital (Hydrogen) B. s orbital --- p orbital (Hydrogen Chloride) C. p orbital --- p orbital (Chlorine) Pi Bonding: Occurs when p orbitals overlap sidewards. Occurs when sigma bond has already been formed between two orbitals. There is less overlap between orbitals in a pi bond and therefore it is weaker. Therefore double bond is not twice as strong as a single bond.
|
Comparison of the Properties of Ionic and Covalent Compounds
|
Ionic:
1. High melting and boiling points 2. Usually solid at room temperature 3. Conduct electricity when dissolved in water or molten |
Covalent:
1. Usually have low melting and boiling points 2. Usually liquid or gases at room temperature 3. So not conduct electricity (no ions present) |
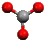
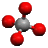
Shapes of Covalent Molecules
Bond Pair: Electrons that make up the covalent bond
Lone Pair: Pair of electrons not involved in a covalent bond
VSEPR: Valence Shell Electron Pair Repulsion Theory, states that since electrons are negatively charged, pairs of electrons will repel each other until they are as far apart as possible, which determines shape of molecule
Lone Pair: Pair of electrons not involved in a covalent bond
VSEPR: Valence Shell Electron Pair Repulsion Theory, states that since electrons are negatively charged, pairs of electrons will repel each other until they are as far apart as possible, which determines shape of molecule
|
|
Electronegativity
ELECTONEGATIVITY: is the relative attraction that an atom in a molecule has for the shared pair of electrons in a covalent bond