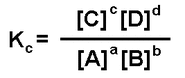Equilibrium
|
Stress Applied |
Reactions Response |
|
Pressure Increased |
Goes to side with lower no. of molecules |
|
Pressure Decreased |
Goes to side with greater no. of molecules |
|
Temperature Increased |
Goes in endothermic direction |
|
Temperature Decreased |
Goes in exothermic direction |
|
Concentration X Increased |
Goes in direction that removes X |
|
Concentration X Decreased |
Goes in direction that forms X |
Pressure change only has an effect if:
- All substances are gases
- Total no. of molecules on left and right are different
Chemical Equilibrium
EQUILIBRIUM: state of dynamic balance where rate of forward reaction equals rate of backwards reaction
- Equilibrium is dynamic as two reactions are still happening
- Reactions at equilibrium are not gone to completion, both reactants and products are present at equilibrium.
Le Chatelier's Principle
LE CHATELIER'S PRINCIPLE: If a stress is applied to a system at equilibrium, the system readjust to relieve the stress
Investigating Le Chatelier's Principle:
CoCl42- + 6H2O ⇌ Co(H2O)62+ + 4Cl- ΔH = -
(blue) (red)
Cr2O72- + H2O ⇌ 2CrO42- + 2H+
(orange) (yellow)
FeCl3 + CNS- ⇌ Fe(CNS)2+ + 3Cl-
(yellow) (red)
- Catalyst has no effect on equilibrium, it just speeds up reaction
Investigating Le Chatelier's Principle:
CoCl42- + 6H2O ⇌ Co(H2O)62+ + 4Cl- ΔH = -
(blue) (red)
- Add conc. HCl (i.e. Cl- ions). Left reaction removes Cl- ions
- Add water. Right reaction removes water
- Heat it. Left reaction removes heat (endothermic)
- Ice bath. Right reaction creates heat (exothermic)
Cr2O72- + H2O ⇌ 2CrO42- + 2H+
(orange) (yellow)
- Add NaOH (i.e. takes H+ ions). Right reaction produces H+
- Add dil. HCl (i.e. H+ ions). Left reaction removes H+ ions
FeCl3 + CNS- ⇌ Fe(CNS)2+ + 3Cl-
(yellow) (red)
- Add dil. HCl (i.e. Cl- ions). Left reaction removes Cl- ions
- Add FeCl3. Right reaction removes FeCl3
Industrial Application of Le Chatelier's Principle
- HABER PROCESS
N2 + 3H2 ⇌ 2NH3 ΔH = - 92.4 kJ mol-1
Ammonia is manufactured by combining nitrogen (from air) and hydrogen from air (using an iron catalyst)
Pressure: High pressure forces reaction to right, therefore increasing ammonia yield. Extremely high pressure are
expensive and dangerous so compromise of 200 atmospheres is used
Temperature: Low temperature favours exothermic reaction so will give higher ammonia yield. However low
temperature decreases rate of reaction. To avoid uneconomically slow process compromise of 500°C is chosen
2. THE CONTACT PROCESS
2SO2 + O2 ⇌ 2SO3 ΔH = - 196 kJ mol-1
Sulfuric Acid is manufactured as above using vandium pentoxide (V2O5) catalyst
Pressure: High pressure increases yield of SO3, but difficulties like high costs and liquefying sulfur dioxide means process
is carried out at only just above atmospheric pressure.
Temperature: Low temperatures forces reaction to right exothermic direction, favouring SO3 production. However,
reaction would be too slow to be economically viable so compromise of 450°C is used
The Equilibrium Constant
- The larger the Kc value, the greater the extent reactions has gone to completion
- Values subbed in are for conc. at equilibrium
- Kc is a constant only if temperature doesn't change
- If equation can be written in different ways, different Kc values will be obtained
Equilibrium constant Kc is when the values for C, D, A and B are the formulas for the molecules, e.g. N2 + H2 = NH3
To calculate Kc fill in the mol/l for the formulas of the molecules
Calculations:
e.g. H2 + I2 ⇌ 2HI
4 moles of H2 mixed with 3 moles I2 and when reaction is at completion, 1 mole I2 was present
e.g. H2 + I2 ⇌ 2HI
4 moles of H2 mixed with 3 moles I2 and when reaction is at completion, 1 mole I2 was present
|
Initially Change Moles at Equilibrium |
H2
4 -2 2 |
I2
3 -2 1 |
¦
¦ ¦ ¦ |
HI
0 +4_ 4 |
Kc = [HI]2 = (4)2_ = 8
[H2][I2] (2)(1)
[H2][I2] (2)(1)
e.g. PCl5 ⇌ PCl3 + Cl2
83.4g of PCl5 were allowed to decompose in a 2l vessel. At equilibrium 15% of the PCl5 remained. Calculate Kc
No. Mol PCl5 initially = Mass in g = 83.4g = 0.4
Mr 208.5g
15% of 0.4 = 0.06 mol at equilibrium
Mr 208.5g
15% of 0.4 = 0.06 mol at equilibrium
|
Initially Change Moles at Equilibrium (2l) Moles in 1l vessel |
PCl5
0.4 -0.34 0.06 0.03 |
¦
¦ ¦ ¦ ¦ |
PCl3
0 +0.34 0.34 0.17 |
Cl2
0 +0.34 0.34 0.17 |
Kc = [PCl3][Cl2] = (0.17)(0.17) = 0.96
[PCl5] (0.03)
[PCl5] (0.03)
e.g. Value for Kc for below reaction was 0.7 at 900°C
CO + H2O ⇌ CO2 + H2
Find mass of CO that would be present at equilibrium if 6 moles of CO are mixed with 6 moles H2O and allowed to react
at 900°C
|
Initially Change Moles at Equilibrium |
CO
6 -x 6-x |
H2O
6 -x 6-x |
¦
¦ ¦ ¦ |
|
Kc = [CO2][H2] = 0.7
[CO][H2O]
x2 = 0.7 = x = √0.7 = 0.837
(6-x)2 (6-x)
=> x = 0.837(6 - x)
=> x = 5.02 - 0.837x
=> x + 0.837x = 5.02
=> 1.837x = 5.02
=> x = 5.02 = 2.73
1.837
No. moles of CO = (6 - x) = (6 - 2.73)
= 3.27
Mass CO = (3.27)(28g)
= 91.56g of CO
[CO][H2O]
x2 = 0.7 = x = √0.7 = 0.837
(6-x)2 (6-x)
=> x = 0.837(6 - x)
=> x = 5.02 - 0.837x
=> x + 0.837x = 5.02
=> 1.837x = 5.02
=> x = 5.02 = 2.73
1.837
No. moles of CO = (6 - x) = (6 - 2.73)
= 3.27
Mass CO = (3.27)(28g)
= 91.56g of CO