The Atom
- All matter is composed of particles (atoms, molecules or ions
- The atom is the basic unit from which all substances are formed
HISTORY OF THE ATOM

John Dalton:
Developed a theory which says that matter is composed of tiny, indivisible particles called atoms that can neither be created nor destroyed
Developed a theory which says that matter is composed of tiny, indivisible particles called atoms that can neither be created nor destroyed
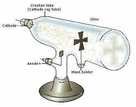
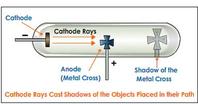
William Crookes:
Showed some form of radiation passes from negative electrode (cathode) to positive electrode (anode) when a current passes through a vacuum tube: he names them cathode rays
Showed some form of radiation passes from negative electrode (cathode) to positive electrode (anode) when a current passes through a vacuum tube: he names them cathode rays

Robert Millikan:
Used the oil drop experiment to measure size of charge on electron. This allowed mass to be calculated. Mass was found to be much smaller than that of any atom. Confirmed atom itself is made up of smaller particles
Used the oil drop experiment to measure size of charge on electron. This allowed mass to be calculated. Mass was found to be much smaller than that of any atom. Confirmed atom itself is made up of smaller particles
|
Ernest Rutherford:
Rutherford bombarded a gold foil (leaf) with alpha particles (positively charged). He used a detector (fluorescent screen) to determine what happened to the alpha particles after they had struck the gold foil. He found that:
From this information Rutherford concluded that
|
|

James Chadwick:
By bombarding a sample of beryllium with alpha particles, Chadwick brought about the release of neutral particles with about the same mass as that of the proton. He called these neutral particles 'neutrons'.
By bombarding a sample of beryllium with alpha particles, Chadwick brought about the release of neutral particles with about the same mass as that of the proton. He called these neutral particles 'neutrons'.