The Periodic Table
The Elements - Historical Background
Ancient Greeks: believed substances composed of earth, wind, air and fire
Robert Boyle: defined element as a substance that cannot be split into simpler substances by chemical means
Humphry Davy: Used electrolysis to isolate potassium from potassium hydroxide, sodium from sodium hydroxide
Henry Moseley: Devised method to determine number of protons in nucleus and called it atomic number. Used to arrange modern periodic table
Robert Boyle: defined element as a substance that cannot be split into simpler substances by chemical means
Humphry Davy: Used electrolysis to isolate potassium from potassium hydroxide, sodium from sodium hydroxide
Henry Moseley: Devised method to determine number of protons in nucleus and called it atomic number. Used to arrange modern periodic table
History of the Periodic Table
Dobereiner: believed elements could be arranged in groups of three and called them triads e.g. CL, Br, and I. The elements in the triad had similar properties and the atomic weight of the middle element was approximate average of the atomic weights of the other two
John Newland: arranged elements in order of increasing atomic weight with every eighth element had similar properties to the first one. He proposed Law of Octaves. He developed a table but it had limited applications, as some elements were not discovered
Dmitri Mendeleev: arranged elements in order of increasing atomic weight and produced a table similar to Newlands, but he left blank spaces assuming elements were to be discovered. Made predictions about elements that proved to be extremely accurate. He also reversed the order of some elements so the would fit.
John Newland: arranged elements in order of increasing atomic weight with every eighth element had similar properties to the first one. He proposed Law of Octaves. He developed a table but it had limited applications, as some elements were not discovered
Dmitri Mendeleev: arranged elements in order of increasing atomic weight and produced a table similar to Newlands, but he left blank spaces assuming elements were to be discovered. Made predictions about elements that proved to be extremely accurate. He also reversed the order of some elements so the would fit.
The Modern Periodic Table
PERIODIC LAW: when elements are arranged in order of increasing atomic number, the properties of the elements of the elements vary periodically
|
Mendeleev's Table:
1. Elements in order of atomic weight 2. Gaps present 3. Transition metals in separate block |
Modern Table:
1. Elements in order of atomic number 2. No gaps present 3. Trasnition metals in seperate block |
Making Use of the Periodic Table
a) Atomic Numbers, Mass Numbers and Isotopes
ATOMIC NUMBER: of an atom is the number of protons in the nucleus of that atom
MASS NUMBER: of an element is the sum of the numbers of protons and neutrons in the nucleus of an atom of that element
ISOTOPES: are atoms of the same element (i.e. they have the same atomic number) but have different mass numbers due to the different number of neutrons in the nucleus
e.g. Carbon has three isotopes carbon-12, carbon-13, carbon-14. The mass number of the elements gives the average mass of the element taking the relative abundances of its isotopes into account
b) Relative Atomic Mass - The Mass Spectometer
RELATIVE ATOMIC MASS: is the average of the mass numbers of the isotopes of the element as they occur naturally, taking their abundancies into account and compared with 1/12th of the mass of the carbon-12 isotopes
ATOMIC NUMBER: of an atom is the number of protons in the nucleus of that atom
MASS NUMBER: of an element is the sum of the numbers of protons and neutrons in the nucleus of an atom of that element
ISOTOPES: are atoms of the same element (i.e. they have the same atomic number) but have different mass numbers due to the different number of neutrons in the nucleus
e.g. Carbon has three isotopes carbon-12, carbon-13, carbon-14. The mass number of the elements gives the average mass of the element taking the relative abundances of its isotopes into account
b) Relative Atomic Mass - The Mass Spectometer
RELATIVE ATOMIC MASS: is the average of the mass numbers of the isotopes of the element as they occur naturally, taking their abundancies into account and compared with 1/12th of the mass of the carbon-12 isotopes
|
Vaporisation: sample must be converted to gas. Inlet chamber is evacuated and this will change a liquid into a vapour
Ionisation: sample is bombarded with electrons. This knocks electrons out of the atoms or molecules and coverts sample to positively charged ions Acceleration: positive ions are accelerated towards a negatively charged plate. A thin beam of ions passes through a hole in this plate Separation: a magnetic field is used to deflect beam of ions. Lighter particles will be deflected more than heavier ones, and so the particles are separated on the basis of their mass Detection: the ions are electronically detected then the signal is amplified and recorded as a mass spectrum |
Uses of Mass Spectrometry:
The principle of mass spectrometry is that charged particles moving in a magnetic field are separated according to the masses of the particles
- Measure relative atomic mass/ relative molecular mass
- Measure the relative abundance of isotopes
- Identify compounds e.g. drug testing and in analysis of gases from waste dumps
The principle of mass spectrometry is that charged particles moving in a magnetic field are separated according to the masses of the particles
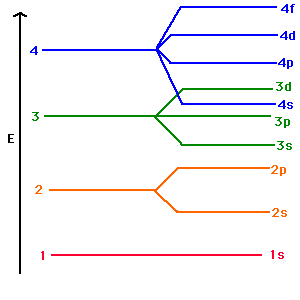
c) Writing Electronic Configurations
|
s sub-level hold 2 electrons
p sub-level holds 6 electrons d sub-level holds 10 electrons n=1 has one sub level n=2 has two sub levels n=3 has three sub levels AUFBAU PRINCIPLE: when building up the electron configuration of an atom in its ground state, the electrons occupy lowest available energy level Sub level 4s is at a lower energy than 3d so electrons occupy it first For example: Nitrogen (7 electrons) = 1s2, 2s2, 2p3 Sulfur (16 electrons) = 1s2, 2s2, 2p6, 3s2, 3p4 Nickel (28 electrons) = 1s2, 2s2, 2p6, 3s2, 3p6, 4s2, 3d8 |
Exceptions:
Electronic Configuration of Ions:
- Cr = 1s2, 2s2, 2p6, 3s2, 3p6, 4s1, 3d5
- Cu = 1s2, 2s2, 2p6, 3s2, 3p6, 4s1, 3d10
Electronic Configuration of Ions:
- An ion with a negative charge has gained one or more electrons
- If an ion has a positive charge it has lost one of more electrons
Arrangement of Electrons in Orbitals of Equal Energy
PAULI EXCLUSION PRINCIPLE: no more that two electrons can occupy an orbital and they must have opposite spin
HUND'S RULE: when two or more orbitals of equal energy are available, electrons occupy them singly before filling them in pairs
e.g. Sulfur (16 electrons) = 1s2, 2s2, 2p6, 3s2, 3px2, 3py1, 3pz1
HUND'S RULE: when two or more orbitals of equal energy are available, electrons occupy them singly before filling them in pairs
e.g. Sulfur (16 electrons) = 1s2, 2s2, 2p6, 3s2, 3px2, 3py1, 3pz1